Abstract
PIs resistance is a major challenge for multiple myeloma (MM). The bone marrow microenvironment facilitates crucial interactions between the myeloma cells and mesenchymal stem cells (MSCs) that permit MM to survive and proliferate progression. Exosomes are involved in intercellular communication, and in this study we investigated how the transfer of exosomic PMSA3 (encodes proteasome subunit α7) and lncPSMA3-AS1 from MSCs to MM cells affected proteasome inhibitors resistance (Figure 1).
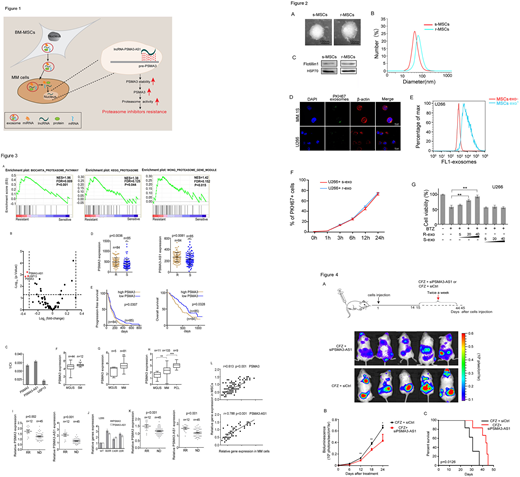
We firstly underscored that exosomes derived from r-MSCs (MSCs derived from bortezomib-resistant patients), but not from s-MSCs (MSCs derived from bortezomib-resistant patients) reduced the proteasome inhibitors sensitivity in MM cells (Figure 2).
To further elucidate mechanisms of Proteasome inhibitors (PIs) resistance, we retrieved a database containing gene expression profile of 169 myeloma cases with clinical response and disease prognosis (GSE9782). The analysis of this dataset showed that the mRNA levels of PSMA3 and PSMA3-AS1 in CD138+ cells are upregulated in bortezomib-resistant patients (Figure 3A-3D). Moreover, Kaplan-Meier analysis showed that high PSMA3 levels in CD138+ MM cells were correlated with reduced progression-free survival (PFS) (p = 0.0307) and overall survival (OS) (p = 0.0328) (Figure 3E). Cox proportional hazards regression analysis further demonstrated that high PSMA3 was an independent prognostic factor for MM patients with bortezomib therapy in a multivariate analysis (p = 0.0013, HR = 1.3104, 95%CI = 1.1113-1.545). Further analysis of Oncomine data showed that the PSMA3 levels appeared a progressive increase in MGUS, SM, MM and PCL (Figure 3F-3H). Similarly, our PIs resistant models (U266BR, U266CR, U266IR, MM.1SBR, MM.1SCR, MM.1SIR) consistently displayed up-regulation of PSMA3 and PSMA3-AS1 expression (Figure 3J). Consistent with this previously published study, our clinical data showed that the mRNA levels of PSMA3 and PSMA3-AS1 are upregulated in CD138+ MM cells derived from bortezomib resistant patients relative to those from bortezomib sensitive patients (Figure 3I). In addition, r-MSCs had increased expression of PSMA3 and PSMA3-AS1 compared to s-MSCs (Figure 3K). Moreover, the expression of PSMA3 and PSMA3-AS1 in MSCs were positively correlated with that in CD138+ myeloma cells (Figure 3L). These data suggested that high levels of PSMA3 and PSMA3-AS1 were correlated with proteasome inhibitors resistance in MM.
We further identified that PSMA3 and PSMA3-AS1 in MSCs could be incorporated into exosomes and transmitted to myeloma cells, thus promoting PIs resistance (Figure not shown).
PSMA3-AS1 was capable of forming an RNA duplex with PSMA3 pre-mRNA at overlapping regions and this duplex transcriptionally promoted PSMA3 expression by increasing its stability, conferring bortezomib resistance to myeloma cells (Figure not shown).
To evaluate the therapeutic potential of PSMA3-AS1 in MM in vivo, bioluminescent MM models (U266-luc), which recapitulates the clinical sequelae, anatomic distribution of MM lesions, and hallmark bone pathophysiology observed in MM patients were established. Intravenously administered siPSMA3-AS1 was found to be effective in increasing bortezomib sensitive (Figure 4).
Moreover, circulating exosomal PSMA3 and PSMA3-AS1 derived from the plasma of MM patients were significantly associated with both progression-free survival (PFS) and overall survival (OS) in the univariate analysis, and were still statistically significant after adjusting for the international staging system (ISS) and several other clinical variables in the multivariate analysis (Figure not shown).
In summary, our results indicated a unique role of exosomic lncPSMA3-AS1 in transferring proteasome inhibitors resistance from MSCs to MM cells, through a novel exosomic lncPSMA3-AS1/PSMA3 signaling pathway. Exosomic PSMA3 and PSMA3-AS1 may serve as a potential therapeutic target for proteasome inhibitors resistance and a prognostic predictor for clinical response.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal